Will Acetone Work For Cleaning Enamel From Paint Brushes?
| |||
| |||
 | |||
| Names | |||
|---|---|---|---|
| IUPAC proper noun Acetone[6] | |||
| Preferred IUPAC name Propan-2-one[7] | |||
Other names
| |||
| Identifiers | |||
| CAS Number |
| ||
| 3D model (JSmol) |
| ||
| 3DMet |
| ||
| Beilstein Reference | 635680 | ||
| ChEBI |
| ||
| ChEMBL |
| ||
| ChemSpider |
| ||
| ECHA InfoCard | 100.000.602 | ||
| EC Number |
| ||
| Gmelin Reference | 1466 | ||
| KEGG |
| ||
| MeSH | Acetone | ||
| PubChem CID |
| ||
| RTECS number |
| ||
| UNII |
| ||
| UN number | 1090 | ||
| CompTox Dashboard (EPA) |
| ||
| InChI
| |||
| SMILES
| |||
| Properties | |||
| Chemical formula | C 3 H 6 O | ||
| Molar mass | 58.080 g·mol−1 | ||
| Appearance | Colourless liquid | ||
| Odour | Pungent, irritating, floral, cucumber similar | ||
| Density | 0.7845 grand/cm3 (25 °C) | ||
| Melting betoken | −94.7 °C (−138.5 °F; 178.5 Thousand)[12] | ||
| Boiling point | 56.05 °C (132.89 °F; 329.20 1000)[12] | ||
| Solubility in h2o | Miscible | ||
| Solubility | Miscible in benzene, diethyl ether, methanol, chloroform, ethanol[8] | ||
| log P | −0.16[9] | ||
| Vapor pressure |
| ||
| Acidity (pThousand a) |
| ||
| Magnetic susceptibility (χ) | −33.78·10−6 cmiii/mol | ||
| Refractive index (n D) | 1.3588 (V D = 54.46) | ||
| Viscosity | 0.295mPa·due south (25 °C)[eight] | ||
| Structure | |||
| Coordination geometry | Trigonal planar at C2 | ||
| Molecular shape | Dihedral at C2 | ||
| Dipole moment | 2.91 D | ||
| Thermochemistry | |||
| Heat capacity (C) | 125.45J/(mol·Grand) | ||
| Std molar | 200.ivJ/(mol·M) | ||
| Std enthalpy of | (−250.03)–(−248.77)kJ/mol | ||
| Std enthalpy of | −i.772MJ/mol | ||
| Hazards | |||
| GHS labelling: | |||
| Pictograms |   | ||
| Signal word | Danger | ||
| Run a risk statements | H225, H319, H336, H373 | ||
| Precautionary statements | P210, P235, P260, P305+P351+P338 | ||
| NFPA 704 (burn diamond) | i 3 0 | ||
| Flash point | −20 °C (−iv °F; 253 Grand) | ||
| Autoignition | 465 °C (869 °F; 738 K) | ||
| Explosive limits | ii.6–12.8%[13] | ||
| Threshold limit value (TLV) | 1185mg/m3 (TWA), 2375mg/thousand3 (STEL) | ||
| Lethal dose or concentration (LD, LC): | |||
| LD50 (median dose) |
| ||
| LC50 (median concentration) | 20,702ppm (rat, eight h)[14] | ||
| LCLo (lowest published) | 45,455ppm (mouse, one h)[fourteen] | ||
| NIOSH (U.s.a. health exposure limits): | |||
| PEL (Permissible) | chiliadppm (2400mg/one thousand3)[3] | ||
| REL (Recommended) | TWA 250ppm (590mg/m3)[3] | ||
| IDLH (Firsthand danger) | 2500ppm[3] | ||
| Related compounds | |||
| Related compounds |
| ||
| Supplementary data page | |||
| Acetone (data page) | |||
| Except where otherwise noted, information are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |||
| | |||
| Infobox references | |||
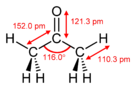
Acetone, propanone or dimethyl ketone, is an organic compound with the formula (CH3)twoCO.[15] It is the simplest and smallest ketone. It is a colourless, highly volatile and combustible liquid with a feature pungent olfactory property.
Acetone is miscible with water and serves every bit an important organic solvent in its ain correct, in industry, habitation, and laboratory. About 6.7 million tonnes were produced worldwide in 2010, mainly for use every bit a solvent and product of methyl methacrylate (and from that PMMA) likewise equally bisphenol A.[16] [17] It is a common building block in organic chemical science. Familiar household uses of acetone are as the active ingredient in blast smoothen remover and as paint thinner. It has volatile organic compound (VOC) exempt condition in the The states.[18]
Acetone is produced and tending of in the homo body through normal metabolic processes. It is normally nowadays in blood and urine. People with diabetic ketoacidosis produce it in larger amounts. Reproductive toxicity tests show that it has low potential to cause reproductive problems. Ketogenic diets that increment ketone bodies (acetone, β-hydroxybutyric acid and acetoacetic acid) in the blood are used to counter epileptic attacks in infants and children who suffer from refractory epilepsy.[19]
History [edit]
Acetone was start produced by Andreas Libavius in 1606 past distillation of Atomic number 82(Two) acetate.[twenty] [21]
In 1832, French chemist Jean-Baptiste Dumas and German language chemist Justus von Liebig determined the empirical formula for acetone.[22] [23] In 1833, the French pharmacist Antoine Bussy named acetone by adding the suffix -one to the stem of the corresponding acid (viz, acerb acid).[24] By 1852, English chemist Alexander William Williamson realized that acetone was methyl acetyl;[25] the following year, the French chemist Charles Frédéric Gerhardt concurred.[26] In 1865, the High german chemist Baronial Kekulé published the modern structural formula for acetone.[27] [28] Johann Josef Loschmidt had presented the structure of acetone in 1861,[29] but his privately published booklet received little attention. During Earth War I, Chaim Weizmann developed the process for industrial production of acetone (Weizmann Process).[30]
Product [edit]
In 2010, the worldwide product chapters for acetone was estimated at 6.7 million tonnes per year.[31] With 1.56 million tonnes per year, the U.s. had the highest production capacity,[32] followed past Taiwan and mainland Communist china. The largest producer of acetone is INEOS Phenol, owning 17% of the globe'south capacity, with also significant chapters (7–viii%) by Mitsui, Sunoco and Vanquish in 2010.[31] INEOS Phenol also owns the globe'due south largest production site (420,000 tonnes/annum) in Beveren (Belgium). Spot toll of acetone in summer 2011 was 1100–1250 USD/tonne in the United States.[33]
Current method [edit]
Acetone is produced straight or indirectly from propylene. Approximately 83% of acetone is produced via the cumene process;[17] as a issue, acetone product is tied to phenol product. In the cumene procedure, benzene is alkylated with propylene to produce cumene, which is oxidized by air to produce phenol and acetone:
Other processes involve the direct oxidation of propylene (Wacker-Hoechst process), or the hydration of propylene to requite 2-propanol, which is oxidized (dehydrogenated) to acetone.[17]
Older methods [edit]
Previously, acetone was produced by the dry out distillation of acetates, for instance calcium acetate in ketonic decarboxylation.
- Ca(CH3COO)2 → CaO(s) + COii(g) + (CHthree)2CO (v)
After that time, during World War I, acetone was produced using acetone-butanol-ethanol fermentation with Clostridium acetobutylicum bacteria, which was developed by Chaim Weizmann (after the start president of Israel) in order to assistance the British war effort,[17] in the preparation of Cordite.[34] This acetone-butanol-ethanol fermentation was eventually abandoned when newer methods with better yields were constitute.[17]
Chemic backdrop [edit]
Keto/enol tautomerism [edit]
Similar about ketones, acetone exhibits the keto–enol tautomerism in which the nominal keto construction (CH
three )
two C=O of acetone itself is in equilibrium with the enol isomer (CH
3 )C(OH)=(CH
2 ) (prop-1-en-2-ol). In acetone vapor at ambient temperature, simply 2.four×10 −7 % of the molecules are in the enol form.[35] All the same the enol course is chemically important in some chemic reactions.[ commendation needed ]
Aldol condensation [edit]
In the presence of suitable catalysts, ii acetone molecules also combine to grade the chemical compound diacetone alcohol (CH
three )C=O(CH
2 )C(OH)(CH
iii )
2 , which on dehydration gives mesityl oxide (CH
iii )C=O(CH)=C(CH3)
two . This product tin can further combine with another acetone molecule, with loss of another molecule of water, yielding phorone and other compounds.[ commendation needed ]
Polymerisation [edit]
1 might expect acetone to besides form polymers and (maybe cyclic) oligomers of two types. In one type, units could be acetone molecules linked by ether bridges –O– derived by from the opening of the double bond, to requite a polyketal-like (PKA) chain [–O–C(CH
3 )
2 –] n . The other blazon could exist obtained through repeated aldol condensation, with one molecule of water removed at each step, yielding a poly(methylacetylene) (PMA) concatenation [–CH=C(CH
3 )–] northward .[36]
PKA type [edit]
The conversion of acetone to a polyketal (PKA) would be coordinating to the formation of paraformaldehyde from formol, and of trithioacetone from thioacetone. In 1960, Kargin, Kabanov and others observed that the thermodynamics of this process is unfavourable for liquid acetone, then that it (unlike thioacetone and formol) is not expected to polymerise spontaneously, even with catalysts. However, they observed that the thermodynamics became favourable for crystalline solid acetone at the melting point (−96 °C). They claimed to have obtained such a polymer (a white elastic solid, soluble in acetone, stable for several hours at room temperature) by depositing vapor of acetone, with some magnesium equally a catalyst, onto a very cold surface.[37]
In 1962, Wasaburo Kawai reported the synthesis of a similar product, from liquid acetone cooled to −70 to −78 °C, using n-butyl lithium or triethylaluminium equally catalysts. He claimed that the infrared absorption spectrum showed the presence of –O– linkages simply no C=O groups.[38] However, alien results were obtained later by other investigators.[36]
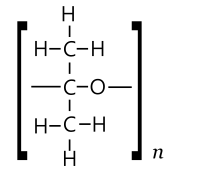
Structure of possible acetone polymer
PMA type [edit]
The PMA type polymers of acetone would exist equivalent to the product of polymerisation of propyne, except for a keto finish group.[36]
Biochemistry [edit]
Biosynthesis [edit]
Small amounts of acetone are produced in the trunk past the decarboxylation of ketone bodies. Certain dietary patterns, including prolonged fasting and loftier-fat low-carbohydrate dieting, tin produce ketosis, in which acetone is formed in body tissue. Sure health conditions, such as alcoholism and diabetes, can produce ketoacidosis, uncontrollable ketosis that leads to a sharp, and potentially fatal, increment in the acidity of the claret. Since it is a byproduct of fermentation, acetone is a byproduct of the distillery manufacture.[ commendation needed ]
Acetone can be produced from the oxidation of ingested isopropanol, or from the spontaneous/enzymatic breakup of acetoacetate (a ketone torso) in ketotic individuals.[ citation needed ]
Metabolism [edit]
Although some biochemistry textbooks and current research publications[39] indicate that acetone cannot be metabolized, there is evidence to the contrary. It can and then be metabolized either past CYP2E1 via methylglyoxal to D-lactate and pyruvate, and ultimately glucose/energy, or by a dissimilar pathway via propylene glycol to pyruvate, lactate, acetate (usable for energy) and propionaldehyde.[40] [41] [42]
Uses [edit]
Industrial [edit]
Well-nigh a third of the globe's acetone is used as a solvent, and a quarter is consumed equally acetone cyanohydrin, a precursor to methyl methacrylate.[16]
Solvent [edit]
Acetone is a practiced solvent for many plastics and some constructed fibers. Information technology is used for thinning polyester resin, cleaning tools used with information technology, and dissolving two-role epoxies and superglue before they harden. It is used equally ane of the volatile components of some paints and varnishes. As a heavy-duty degreaser, it is useful in the grooming of metal prior to painting or soldering, and to remove rosin flux after soldering (to forestall adhesion of dirt and electrical leakage and maybe corrosion or for cosmetic reasons), although it attacks many electronic components (for example polystyrene capacitors) so it is unsuitable for cleaning many circuit boards.[ commendation needed ]
Acetylene carrier [edit]
Although itself flammable, acetone is used extensively as a solvent for the safe transportation and storage of acetylene, which cannot be safely pressurized as a pure compound. Vessels containing a porous fabric are first filled with acetone followed by acetylene, which dissolves into the acetone. One litre of acetone can dissolve around 250 litres of acetylene at a pressure of 10 bars (1.0 MPa).[43] [44]
Chemical intermediate [edit]
Acetone is used to synthesize methyl methacrylate. It begins with the initial conversion of acetone to acetone cyanohydrin:
- (CH3)2CO + HCN → (CH3)2C(OH)CN
In a subsequent stride, the nitrile is hydrolyzed to the unsaturated amide, which is esterified:
- (CH3)2C(OH)CN + CH3OH → CH2=C(CH3)CO2CHthree + NHiii
The third major use of acetone (about 20%)[16] is synthesizing bisphenol A. Bisphenol A is a component of many polymers such as polycarbonates, polyurethanes, and epoxy resins. The synthesis involves the condensation of acetone with phenol:
- (CHthree)2CO + ii C6HfiveOH → (CH3)2C(Chalf-dozenH4OH)2 + H2O
Many millions of kilograms of acetone are consumed in the production of the solvents methyl isobutyl alcohol and methyl isobutyl ketone. These products arise via an initial aldol condensation to give diacetone alcohol.[17]
- 2 (CH3)2CO → (CH3)iiC(OH)CHiiC(O)CHiii
Condensation with acetylene gives 2-methylbut-3-yn-ii-ol, precursor to synthetic terpenes and terpenoids.[ citation needed ]
Laboratory [edit]
Chromatography [edit]

Spectroscopy techniques are useful when the sample being tested is pure, or a very common mixture. When an unknown mixture is being analyzed information technology must exist broken downward into its individual parts. Chromatography techniques tin be used to suspension autonomously mixtures into their components allowing for each function to be analyzed separately.
Thin layer chromatography (TLC) is a quick alternative to more complex chromatography methods. TLC tin can be used to analyze inks and dyes past extracting the individual components.[45] This can be used to investigate notes or fibers left at the scene since each visitor's product is slightly different and those differences tin can exist seen with TLC. The only limiting factor with TLC analysis is the necessity for the components to be soluble in whatever solution is used to carry the components upward the analysis plate.[45] This solution is called the mobile phase.[45] The forensic chemist can compare unknowns with known standards past looking at the distance each component travelled.[45] This distance, when compared to the starting bespeak, is known equally the retention factor (Rf) for each extracted component.[45] If each Rf value matches a known sample, that is an indication of the unknown'due south identity.[45]
Loftier-performance liquid chromatography can exist used to extract individual components from a mixture dissolved in a solution. HPLC is used for nonvolatile mixtures that would non be suitable for gas chromatography.[46] This is useful in drug analysis where the pharmaceutical is a combination drug since the components would carve up, or elute, at different times allowing for the verification of each component.<[47] The eluates from the HPLC column are then fed into various detectors that produce a pinnacle on a graph relative to its concentration every bit it elutes off the column. The nearly mutual type of detector is an ultraviolet-visible spectrometer as the most mutual item of interest tested with HPLC, pharmaceuticals, take UV absorbance.<[48]
Gas chromatography (GC) performs the aforementioned function as liquid chromatography, just information technology is used for volatile mixtures. In forensic chemical science, the nigh common GC instruments use mass spectrometry as their detector.[49] GC-MS tin be used in investigations of arson, poisoning, and explosions to make up one's mind exactly what was used. In theory, GC-MS instruments can observe substances whose concentrations are in the femtogram (10 −15 ) range.[fifty] However, in practice, due to signal-to-noise ratios and other limiting factors, such as the age of the individual parts of the instrument, the practical detection limit for GC-MS is in the picogram (10 −12 ) range.[51] GC-MS is also capable of quantifying the substances information technology detects; chemists tin can utilize this information to determine the effect the substance would have on an individual. GC-MS instruments need around 1,000 times more of the substance to quantify the amount than they demand merely to detect information technology; the limit of quantification is typically in the nanogram (10 −9 ) range.[51]
Chemical inquiry [edit]
In the laboratory, acetone is used as a polar, aprotic solvent in a variety of organic reactions, such equally SN2 reactions. The utilize of acetone solvent is critical for the Jones oxidation. It does not form an azeotrope with water (see azeotrope tables).[52] Information technology is a common solvent for rinsing laboratory glassware because of its low cost and volatility. Despite its mutual use as a supposed drying agent, it is not constructive except by bulk deportation and dilution. Acetone tin exist cooled with dry out ice to −78 °C without freezing; acetone/dry ice baths are commonly used to conduct reactions at low temperatures. Acetone is fluorescent under ultraviolet light, and its vapor tin can be used as a fluorescent tracer in fluid menstruum experiments.[53]
Acetone is used to precipitate proteins.[54] Alternatives for protein precipitation are trichloroacetic acid or ethanol.[ citation needed ]
Cleaning [edit]
Low-grade acetone is also ordinarily used in academic laboratory settings as a glassware rinsing agent for removing residue and solids before a final wash.[55] Acetone leaves a small amount of residue on a surface when stale that is harmful to surface samples.[ citation needed ]
Low-temperature bath [edit]
A mixture of acetone and dry ice is a popular cooling bath that maintains a temperature of −78 °C equally long as there is some dry ice left.[56]
Histology [edit]
Acetone is used in the field of pathology to find lymph nodes in fatty tissues for tumor staging (such as looking for lymph nodes in the fatty surrounding the intestines).[57] This helps deliquesce the fat, and hardens the nodes, making finding them easier.[58]
Acetone also used for destaining microscope slides of certain stains.[59]
Lewis base of operations properties [edit]
Acetone is a weak Lewis base that forms adducts with soft acids similar I2 and hard acids like phenol. Acetone also forms complexes with divalent metals.[60] [61]
Medical [edit]
Drug solvent and excipient [edit]
Acetone is used as a solvent past the pharmaceutical industry and as a denaturant in denatured alcohol.[62] Acetone is also nowadays as an excipient in some pharmaceutical drugs.[63]
Skin defatting [edit]
Dermatologists utilise acetone with booze for acne treatments to chemically peel dry peel. Common agents used today for chemical peeling are salicylic acid, glycolic acid, xxx% salicylic acrid in ethanol, and trichloroacetic acid (TCA). Prior to chemexfoliation, the skin is cleaned and excess fat removed in a process called defatting. Acetone, Septisol, or a combination of these agents is commonly used in this process.[ citation needed ]
Anticonvulsant [edit]
Acetone has been shown to have anticonvulsant furnishings in brute models of epilepsy, in the absence of toxicity, when administered in millimolar concentrations.[64] It has been hypothesized that the high-fat depression-carbohydrate ketogenic diet used clinically to control drug-resistant epilepsy in children works past elevating acetone in the brain.[64] Because of their higher energy requirements, children accept higher acetone product than most adults – and the younger the child, the higher the expected production. This indicates that children are non uniquely susceptible to acetone exposure. External exposures are small compared to the exposures associated with the ketogenic diet.[65]
Domestic and other niche uses [edit]
Acetone is oft the main component in cleaning agents such every bit nail polish and superglue removers. It will attack some plastics, still.[ citation needed ]
Brand-up artists use acetone to remove skin adhesive from the netting of wigs and mustaches by immersing the item in an acetone bath, so removing the softened glue residue with a stiff brush.[ citation needed ]
Acetone is often used for vapor polishing of printing artifacts on 3D-printed models printed with ABS plastic. The technique, chosen acetone vapor bath smoothing, involves placing the printed role in a sealed chamber containing a small corporeality of acetone, and heating to effectually 80 degrees Celsius for ten minutes. This creates a vapor of acetone in the container. The acetone condenses evenly all over the role, causing the surface to soften and liquefy. Surface tension then smooths the semi-liquid plastic. When the part is removed from the bedchamber, the acetone component evaporates leaving a glassy-smooth function gratis of striation, patterning, and visible layer edges, common features in untreated 3D printed parts.[66]
Acetone efficiently removes felt-tipped pen marks from drinking glass and metals.
Safety [edit]
Flammability [edit]
The most chancy property of acetone is its extreme flammability. In small amounts Acetone burns with a dull blue flame, in larger amounts the evaporation of fuel causes incomplete combustion and a bright yellow flame. At temperatures greater than acetone'south flash point of −20 °C (−iv °F), air mixtures of between 2.5% and 12.8% acetone, by book, may explode or cause a wink burn down. Vapors can flow along surfaces to distant ignition sources and flash dorsum. Static discharge may also ignite acetone vapors, though acetone has a very high ignition initiation energy indicate and therefore accidental ignition is rare. Even pouring or spraying acetone over red-glowing coal volition not ignite information technology, due to the high concentration of vapour and the cooling effect of evaporation of the liquid.[67] It auto-ignites at 465 °C (869 °F). Auto-ignition temperature is too dependent upon the exposure time, thus at some tests it is quoted as 525 °C. Also, industrial acetone is likely to contain a small corporeality of water which also inhibits ignition.[ commendation needed ]
Acetone peroxide [edit]
When oxidized, acetone forms acetone peroxide as a byproduct, which is a highly unstable, main high explosive chemical compound. It may be formed accidentally, e.g. when waste matter hydrogen peroxide is poured into waste solvent containing acetone. Due to its instability, it is rarely used, despite its simple chemic synthesis.[ commendation needed ]
Toxicity [edit]
Acetone has been studied extensively and is believed to exhibit just slight toxicity in normal apply. At that place is no strong evidence of chronic health effects if basic precautions are followed.[68] It is more often than not recognized to accept low acute and chronic toxicity if ingested and/or inhaled.[69] Acetone is not currently regarded as a carcinogen, a mutagenic chemical nor a concern for chronic neurotoxicity effects.[67]
Acetone can be found as an ingredient in a variety of consumer products ranging from cosmetics to candy and unprocessed foods. Acetone has been rated as a more often than not recognized every bit safe (GRAS) substance when present in beverages, baked foods, desserts, and preserves at concentrations ranging from 5 to 8 mg/L.[69]
Acetone is however an irritant, causing mild skin irritation and moderate to severe eye irritation. At high vapor concentrations, it may depress the cardinal nervous system like many other solvents.[70] Acute toxicity for mice by ingestion (LDl) is 3 yard/kg, and by inhalation (LCfifty) is 44 g/m3 over 4 hours.[71]
EPA Nomenclature [edit]
In 1995, the United States Ecology Protection Agency (EPA) removed acetone from the list of "toxic chemicals" maintained under Section 313 of the Emergency Planning and Customs Right to Know Act (EPCRA). In making that decision, EPA conducted an extensive review of the available toxicity information on acetone and found that acetone "exhibits astute toxicity only at levels that greatly exceed releases and resultant exposures", and farther that acetone "exhibits low toxicity in chronic studies".[ commendation needed ]
- Genotoxicity. Acetone has been tested in more than two dozen in vitro and in vivo assays. These studies point that acetone is not genotoxic.[ commendation needed ]
- Carcinogenicity. EPA in 1995 ended, "There is currently no prove to propose a concern for carcinogenicity". (EPCRA Review, described in Department three.3). NTP scientists have recommended against chronic toxicity/carcinogenicity testing of acetone considering "the prechronic studies simply demonstrated a very mild toxic response at very high doses in rodents".[ commendation needed ]
- Neurotoxicity and Developmental Neurotoxicity. The neurotoxic potential of both acetone and isopropanol, the metabolic precursor of acetone, accept been extensively studied. These studies demonstrate that although exposure to high doses of acetone may cause transient key nervous system furnishings, acetone is not a neurotoxicant. A guideline developmental neurotoxicity written report has been conducted with isopropanol, and no developmental neurotoxic effects were identified, fifty-fifty at the highest dose tested. (SIAR, pp. 1, 25, 31).
- Environmental. When the EPA exempted acetone from regulation as a volatile organic compound (VOC) in 1995, EPA stated that this exemption would "contribute to the achievement of several of import environmental goals and would support EPA's pollution prevention efforts". 60 Fed. Reg. 31,634 (June 16, 1995). 60 Fed. Reg. 31,634 (June 16, 1995). EPA noted that acetone could exist used equally a substitute for several compounds that are listed as hazardous air pollutants (HAP) under section 112 of the Clean Air Human action.[ citation needed ]
Ecology effects [edit]
Although acetone occurs naturally in the surroundings in plants, trees, volcanic gases, woods fires, and equally a product of the breakdown of body fatty,[72] the bulk of the acetone released into the environment is of industrial origin. Acetone evaporates rapidly, even from water and soil. Once in the temper, it has a 22-day one-half-life and is degraded past UV low-cal via photolysis (primarily into methane and ethane.[73]) Consumption past microorganisms contributes to the dissipation of acetone in soil, animals, or waterways.[72]
The LD50 of acetone for fish is 8.iii g/L of water (or about 1%) over 96 hours, and its environmental one-half-life in water is nearly ane to 10 days. Acetone may pose a significant chance of oxygen depletion in aquatic systems due to the microbial consumption.[74]
[edit]
On 30 July 2015, scientists reported that upon the first touchdown of the Philae lander on comet 67P's surface, measurements by the COSAC and Ptolemy instruments revealed sixteen organic compounds, 4 of which were seen for the start time on a comet, including acetamide, acetone, methyl isocyanate, and propionaldehyde.[75] [76] [77]
References [edit]
- ^ The Merck Index, 15th Ed. (2013), p. 13, Acetone Monograph 65, O'Neil: The Royal Order of Chemistry.(subscription required)
- ^ a b c d Acetone in Linstrom, Peter J.; Mallard, William G. (eds.); NIST Chemical science WebBook, NIST Standard Reference Database Number 69, National Plant of Standards and Applied science, Gaithersburg (MD), http://webbook.nist.gov
- ^ a b c d NIOSH Pocket Guide to Chemic Hazards. "#0004". National Found for Occupational Prophylactic and Health (NIOSH).
- ^ Klamt, Andreas (2005). COSMO-RS: From Breakthrough Chemistry to Fluid Phase Thermodynamics and Drug Design. Elsevier. pp. 92–94. ISBN978-0-444-51994-8.
- ^ Myers, Richard 50. (2007). The 100 Most Important Chemic Compounds: A Reference Guide. Greenwood. pp. four–six. ISBN978-0-313-08057-9.
- ^ ChemSpider lists 'acetone' as a valid, skilful-verified name for what would systematically be called 'propan-2-ane'.
- ^ Nomenclature of Organic Chemical science : IUPAC Recommendations and Preferred Names 2013 (Bluish Volume). Cambridge: The Purple Social club of Chemistry. 2014. p. 723. doi:10.1039/9781849733069-FP001. ISBN978-0-85404-182-4.
- ^ a b Properties of substance: acetone. chemister.ru.
- ^ "acetone". ChemSrc . Retrieved 2018-04-13 .
- ^ Chiang, Yvonne; Kresge, A. Jerry; Tang, Yui S.; Wirz, Jakob (1984). "The pKa and keto-enol equilibrium abiding of acetone in aqueous solution". Journal of the American Chemical Society. 106 (ii): 460–462. doi:10.1021/ja00314a055.
- ^ Bordwell, Frederick Chiliad. (1988). "Equilibrium acidities in dimethyl sulfoxide solution". Accounts of Chemical Inquiry. 21 (12): 456–463. doi:ten.1021/ar00156a004.
- ^ a b Lide, David R. (ed) (2003). CRC Handbook of Chemistry and Physics, 84th Edition. CRC Press. Boca Raton, Florida; Department 3, Physical Constants of Organic Compounds.
- ^ "Working with mod hydrocarbon and oxygenated solvents: a guide to flammability". American Chemical science Quango Solvents Industry Grouping. January 2008. p. 7. Archived from the original on 2009-06-01.
- ^ a b c "Acetone". Immediately Dangerous to Life or Health Concentrations (IDLH). National Institute for Occupational Rubber and Wellness (NIOSH).
- ^ Allen, P.W.; Bowen, H.J.M.; Sutton, L.E.; Bastiansen, O. (1952). "The molecular construction of acetone". Transactions of the Faraday Society. 48: 991. doi:ten.1039/TF9524800991.
- ^ a b c Acetone, World Petrochemicals study, January 2010
- ^ a b c d e f Stylianos Sifniades, Alan B. Levy, "Acetone" in Ullmann's Encyclopedia of Industrial Chemistry, Wiley-VCH, Weinheim, 2005.
- ^ "Update: U.S. EPA Exempt Volatile Organic Compounds". American Coatings Clan. 2018-01-30. Archived from the original on 2021-02-08. Retrieved 2019-03-20 .
- ^ Freeman, JM; Kossoff, EH; Hartman, AL (Mar 2007). "The ketogenic diet: one decade afterward". Pediatrics. 119 (three): 535–43. doi:10.1542/peds.2006-2447. PMID 17332207. S2CID 26629499.
- ^ Libavius, Andreas (1606). Alchymia (in Latin). Frankfurt, (Germany): printed past Joannes Saurius, at the expense of Peter Kopff. p. 123. From p. 123: "QUINTA ESSENTIA PLUMBI. Calcem Saturni macera in aceto stillatitio per biduum in cineribus, & solvetur in acetum tenuis substantia, effunde, reponeque aliud, & sic perge, quoad tota subtilitas est extracta. Coagula acetum collectum in salem. Huic affunde spiritum vini circulatum aut alcalisatum. Circula per dies aliquot, destilla ut essentia exeat per retortam. Hanc edulcora, & est quinta Saturni essentia, quam & saccharum vocant." (Fifth ESSENCE OF Atomic number 82. For ii days I roast [i.e., reflux], in embers, lead steeped in distilled vinegar, and the substance [i.e., lead] is dissolved in the dilute vinegar; pour off [the solution] and render [to the reflux flask] the other [i.east., anything that hasn't dissolved] and so keep until every bit [of pb] has been extracted. Concentrate the collected vinegar into a common salt. To this, cascade in spirit of wine [i.e., ethanol] [that has been] refluxed or treated with alkali. Reflux for a few days; dribble by retort so that the essence [i.e., volatile liquid] is gone. Neutralize this, and [this] is the fifth essence of atomic number 82 and what they telephone call sugar [of lead].)
- ^ "Aceton - Chemgapedia".
- ^ Dumas, J. (1832) "Sur l'esprit pyro-acétique" (On pyro-acetic spirit), Annales de Chimie et de Physique, 2d series, 49 : 208–210.
- ^ Liebig, Justus (1832) "Sur les combinaisons produites par fifty'action du gas oléfiant et fifty'esprit acétique" (On compounds produced by the action of ethylene and acetic spirit), Annales de Chimie et de Physique, 2nd series, 49 : 146–204 (especially 193–204).
- ^ Bussy, Antoine (1833) "De quelques Produits nouveaux obtenus par l'action des Alcalis sur les Corps gras à une haute température" (On some new products obtained by the activeness of alkalies on fatty substances at a loftier temperature), Annales de Chimie et de Physique, 2nd series, 53 : 398–412; come across footnote on pp. 408–409.
- ^ Williamson, A. W. (1852) "On Etherification," Journal of the Chemic Society, 4 : 229–239; (especially pp. 237–239).
- ^ Gerhardt, Charles (1853) "Researches sur les acids organiques anhydres" (Enquiry on anhydrous organic acids), Annales de Chimie et de Physique, 3rd series, 37 : 285–342; see p. 339.
- ^ Kekulé, Auguste (1865) "Sur la constitution des substances aromatiques," Message de la Société chimique de Paris, i : 98–110; (particularly p. 110).
- ^ Kekulé, Auguste (1866) "Untersuchungen über aromatischen Verbindungen" (Investigations into aromatic compounds), Annalen der Chemie und Pharmacie, 137 : 129–196; (specially pp. 143–144).
- ^ Loschmidt, J. (1861) Chemische Studien Vienna, Austria-hungary: Carl Gerold's Sohn.
- ^ Chaim Weizmann. chemistryexplained.com
- ^ a b Greiner, Camara; Funada, C (June 2010). "CEH Marketing Research Study: ACETONE". Chemical Economics Handbook. SRI consulting. Retrieved ii September 2016. (subscription required)
- ^ "Acetone Uses and Marketplace Data". ICIS.com. Oct 2010. Archived from the original on 2009-05-fifteen. Retrieved 2011-03-21 .
- ^ Acetone (United states of america Gulf) Price Report – Chemic pricing information Archived 2013-05-16 at the Wayback Motorcar. ICIS Pricing. Retrieved on 2012-11-26.
- ^ Wittcoff, M.M.; Green, H.A. (2003). Organic chemistry principles and industrial do (1. ed., one. reprint. ed.). Weinheim: Wiley-VCH. p. 4. ISBN3-527-30289-1.
- ^ Hine, Jack; Arata, Kazushi (1976). "Keto-Enol Tautomerism. II. The Calorimetrical Determination of the Equilibrium Constants for Keto-Enol Tautomerism for Cyclohexanone and Acetone". Bulletin of the Chemical Club of Japan. 49 (11): 3089–3092. doi:ten.1246/bcsj.49.3089.
- ^ a b c Cataldo, Franco (1996). "Synthesis of ketonic resins from self-polymerization of acetone, i Action of protic and Lewis acids on acetone". Die Angewandte Makromolekulare Chemie. 236 (1): 1–19. doi:10.1002/apmc.1996.052360101.
- ^ Five. A. Kargin, V. A. Kabanov, Five. P. Zubov, I. M. Papisov (1960): "Polymerisation of acetone". Doklady Akademii Nauk SSSR, volume 134, issue 5, pages 1098–1099. Mi dan24153
- ^ Kawai, Wasaburo (1962). "Polymerization of Acetone". Bulletin of the Chemical Society of Japan. 35 (3): 516A. doi:ten.1246/bcsj.35.516a.
- ^ Vujasinovic, Thousand; Kocar, One thousand; Kramer, K; Bunc, Thou; Brvar, M (2007). "Poisoning with ane-propanol and 2-propanol". Human & Experimental Toxicology. 26 (12): 975–8. doi:10.1177/0960327107087794. PMID 18375643. S2CID 11723110.
- ^ Glew, Robert H (2010). "You Tin can Go There From Here: Acetone, Anionic Ketones and Even-Carbon Fatty Acids can Provide Substrates for Gluconeogenesis". Nig. J. Physiol. Sci. 25: 2–4. Archived from the original on 2013-09-26. Retrieved 2013-09-01 .
- ^ Miller, DN; Bazzano, G (1965). "Propanediol metabolism and its relation to lactic acid metabolism". Ann NY Acad Sci. 119 (3): 957–973. Bibcode:1965NYASA.119..957M. doi:10.1111/j.1749-6632.1965.tb47455.x. PMID 4285478. S2CID 37769342.
- ^ Ruddick, JA (1972). "Toxicology, metabolism, and biochemistry of ane,two-propanediol". Toxicol Appl Pharmacol. 21 (1): 102–111. doi:10.1016/0041-008X(72)90032-4. PMID 4553872.
- ^ Mine Rubber and Health Administration (MSHA) – Safety Hazard Data – Special Hazards of Acetylene Archived 2016-01-22 at the Wayback Car. Msha.gov. Retrieved on 2012-11-26.
- ^ History – Acetylene dissolved in acetone Archived 2015-09-15 at the Wayback Machine. Aga.com. Retrieved on 2012-11-26.
- ^ a b c d eastward f Carlysle, Felicity (2011-07-26). "TLC the Forensic Way". theGIST. Glasgow Insight Into Science & Technology. Archived from the original on July 30, 2016. Retrieved October 10, 2015.
- ^ Picó, Yolanda, ed. (2012). Chemical Analysis of Food: Techniques and Applications. Elsevier. p. 501. ISBN9780123848628 – via Google Books.
- ^ Nikolin, B; Imamović, B; Medanhodzić-Vuk, S; Sober, M (May 2004). "High Performance Liquid Chromatography in Pharmaceutical Analyses". Bosnian Periodical of Basic Medical Sciences. 4 (2): 5–9. doi:10.17305/bjbms.2004.3405. PMC7250120. PMID 15629016.
- ^ Dong, Michael W. (2016). Modern HPLC for Practicing Scientists. John Wiley & Sons. ISBN9780471727897 – via Google Books.
- ^ "A Simplified Guide to Forensic Drug Chemistry" (PDF). National Forensic Scientific discipline Technology Center. Archived from the original (PDF) on March 21, 2016. Retrieved September 24, 2015.
- ^ Fialkov, Alexander; Steiner, Urs; Lehotay, Steven; Amirav, Aviv (Jan xv, 2007). "Sensitivity and Noise in GC-MS: Achieving Low Limits of Detection for Difficult Analytes". International Periodical of Mass Spectrometry. 260 (ane): 31–48. Bibcode:2007IJMSp.260...31F. doi:10.1016/j.ijms.2006.07.002.
- ^ a b Smith, Michael L.; Vorce, Shawn P.; Holler, Justin Thou.; Shimomura, Eric; Magluilo, Joe; Jacobs, Aaron J.; Huestis, Marilyn A. (June 2007). "Mod Instrumental Methods in Forensic Toxicology". Periodical of Analytical Toxicology. 31 (5): 237–253. doi:10.1093/jat/31.5.237. PMC2745311. PMID 17579968.
- ^ What is an Azeotrope?. Solvent—recycling.com. Retrieved on 2012-xi-26.
- ^ Lozano, A.; Yip, B.; Hanson, R.One thousand. (1992). "Acetone: a tracer for concentration measurements in gaseous flows by planar laser-induced fluorescence". Exp. Fluids. 13 (six): 369–376. Bibcode:1992ExFl...xiii..369L. doi:10.1007/BF00223244. S2CID 121060565.
- ^ Simpson, Deborah K.; Beynon, Robert J. (2009). "Acetone atmospheric precipitation of proteins and the modification of peptides". Journal of Proteome Research. ix (1): 444–450. doi:10.1021/pr900806x. ISSN 1535-3907. PMID 20000691.
- ^ "Cleaning Glassware" (PDF). Wesleyan Academy. September 2009. Retrieved July seven, 2016.
- ^ Addison, Ault (1998). Studyguide for Techniques and Experiments for Organic Chemistry. Sausalito, CA. p. 310. ISBN9780935702767.
- ^ Basten, O.; Bandorski, D.; Bismarck, C.; Neumann, M.; Fisseler-Eckhoff, A. (13 December 2009). "Acetonkompression". Der Pathologe (in German). 31 (iii): 218–224. doi:10.1007/s00292-009-1256-7. PMID 20012620.
- ^ Leung, C. A. Westward.; Fazzi, One thousand. E.; Melenhorst, J.; Rennspiess, D.; Grabsch, H. I. (November 2018). "Acetone clearance of mesocolic or mesorectal fat increases lymph node yield and may improve detection of high-risk Stage Ii colorectal cancer patients" (PDF). Colorectal Affliction. 20 (11): 1014–1019. doi:10.1111/codi.14335. PMID 29989291. S2CID 205030844.
- ^ Engbaek, 1000; Johansen, KS; Jensen, ME (February 1979). "A new technique for Gram staining paraffin-embedded tissue". Journal of Clinical Pathology. 32 (2): 187–xc. doi:10.1136/jcp.32.2.187. PMC1145607. PMID 86548.
- ^ Driessen, W.L.; Groeneveld, W.L. (1969). "Complexes with ligands containing the carbonyl group. Part I: Complexes with acetone of some divalent metals containing tetrachloro‐ferrate(Iii) and ‐indate(3) anions". Recueil des Travaux Chimiques des Pays-Bas. 88: 77977–988.
- ^ Kilner, C. A.; Halcrow, Yard. A. (2006). "An unusual example of a linearly coordinated acetone ligand in a six-coordinate iron(II) complex". Acta Crystallographica C. 62 (9): 1107–1109. doi:10.1107/S0108270106028903. PMID 16954630.
- ^ Weiner, Myra L.; Lois A. Kotkoskie (1999). Excipient Toxicity and Safety . p. 32. ISBN978-0-8247-8210-8.
- ^ Inactive Ingredient Search for Approved Drug Products, FDA/Centre for Drug Evaluation and Inquiry
- ^ a b Likhodii SS; Serbanescu I; Cortez MA; Murphy P; Snead OC; Burnham WM (2003). "Anticonvulsant backdrop of acetone, a brain ketone elevated by the ketogenic diet". Ann Neurol. 54 (2): 219–226. doi:10.1002/ana.10634. PMID 12891674. S2CID 3213318.
- ^ American Chemistry Council Acetone Console (September 10, 2003). "Acetone (CAS No. 67-64-1) VCCEP Submission" (PDF). pp. half-dozen, 9. Retrieved 2018-04-xiv .
- ^ "Quality Finish 3D Prints with Acetone". instructables.com
- ^ a b Acetone MSDS. Take a chance.com (1998-04-21). Retrieved on 2012-xi-26.
- ^ Basic Data on Acetone. Ccohs.ca (1999-02-19). Retrieved on 2012-xi-26.
- ^ a b "SIDS Initial Assessment Report: Acetone" (PDF). Environmental Protection Agency. Archived from the original (PDF) on 2014-03-09. Retrieved 2014-09-11 .
- ^ "What are the potential health effects of acetone?". Canadian Centre for Occupational Wellness and Safety. Archived from the original on 2008-10-17. Retrieved 2008-ten-21 .
- ^ Rubber (MSDS) data for propanone Archived 2018-03-xvi at the Wayback Machine. sciencelab.com/msds. Retrieved on 2018-03-xix
- ^ a b Acetone, Bureau for Toxic Substances and Disease Registry ToxFAQs, 1995
- ^ Darwent, B. deB.; Allard, M. J.; Hartman, One thousand. F.; Lange, L. J. (1960). "The Photolysis of Acetone". Journal of Concrete Chemistry. 64 (12): 1847–1850. doi:10.1021/j100841a010.
- ^ "Safety Data Sail Acetone" (PDF). J.M. Loveridge. Archived from the original (PDF) on 2009-03-20. Retrieved 2012-11-26 .
- ^ Jordans, Frank (xxx July 2015). "Philae probe finds evidence that comets can be catholic labs". The Washington Post. Associated Press. Archived from the original on 23 December 2018. Retrieved 30 July 2015.
- ^ "Science on the Surface of a Comet". European Infinite Agency. 30 July 2015. Retrieved 30 July 2015.
- ^ Bibring, J.-P.; Taylor, M.Grand.G.T.; Alexander, C.; Auster, U.; Biele, J.; Finzi, A. Ercoli; Goesmann, F.; Klingehoefer, G.; Kofman, W.; Mottola, S.; Seidenstiker, Thou.J.; Spohn, T.; Wright, I. (31 July 2015). "Philae'south Showtime Days on the Comet – Introduction to Special Issue". Scientific discipline. 349 (6247): 493. Bibcode:2015Sci...349..493B. doi:10.1126/science.aac5116. PMID 26228139.
External links [edit]
| | Wikimedia Commons has media related to Acetone. |
- International Chemical Safety Carte 0087
- NIOSH Pocket Guide to Chemic Hazards
- Acetone Safety Information Sail (SDS)
- Hazardous substances databank entry at the national library of medicine
- SIDS Initial Assessment Report for Acetone from the Organisation for Economic Co-operation and Development (OECD)
- Adding of vapor pressure, liquid density, dynamic liquid viscosity, surface tension of acetone
Source: https://en.wikipedia.org/wiki/Acetone
Posted by: howardkinty1949.blogspot.com







0 Response to "Will Acetone Work For Cleaning Enamel From Paint Brushes?"
Post a Comment